Abstract
Previous studies by our group demonstrate the ability to routinely derive hematopoietic and immune cells from human pluripotent stem cells. Here, we demonstrate the efficient derivation of macrophages from human induced pluripotent stem cells (iPSCs). These macrophages have phenotypic and genotypic characteristics similar to monocytes/macrophages isolated from human peripheral blood. We also demonstrate the ability to polarize these iPSC-derived macrophages (iPSC-Macs) to M1 and M2 populations. Specifically, M1 iPSC-Macs have pro-inflammatory characteristics including expression of CD40 and CD80 on the cell surface, produce increased amounts of TNF-a and IL-6 detected in the supernatant, as well have increased expression of inflammatory cytokines/chemokines (TNF-a, IL-6, IL-1b, IL-12, CCL2, CCL3 and TRAIL) and increased expression of matrix metalloproteases (MMPs).
Function of these iPSC-Macs was initially assessed by phagocytosis of fluorescently-labeled beads. These studies demonstrated both the iPSC-M1 and M2 macrophages efficiently phagocytized these beads, and at similar amounts as their peripheral blood counterparts. Next, we tested the ability of the iPSC-Macs to phagocytize human tumor cells. Using A1847 ovarian tumor cells, we found while the iPSC-Macs alone had limited ability to phagocytize the tumor cells (9%), addition of either an anti-CD47 mAb (41%) or anti-EGFR (41%) lead to markedly increased phagocytosis, with the combination of the 2 antibodies being even better (55% phagocytosis). We then tested iPSC-Macs in vivo against luciferase (luc)-expressing A1847 ovarian cancer cells as a xenograft model in NSG-SGM3 mice that express human IL3, GM-CSF and SCF. Using bioluminescent imaging, we found that the combination of iPSC-Macs with both anti-CD47 and anti-EGFR demonstrated significantly improved anti-tumor activity, with median survival of 75 days, compared to 50-60 days for mice treated with only iPSC-Macs, only mAbs or with iPSC-Macs combined either single mAb.
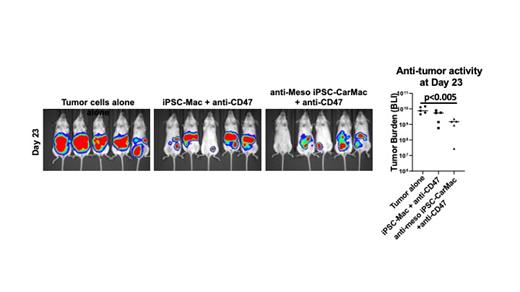
Next, we aimed to use the iPSC platform to produce iPSC-Macs engineered to express chimeric antigen receptors (CARs) to further improve their anti-tumor activity. Here, we developed and tested novel macrophage specific CARs that were stably expressed in undifferentiated iPSCs using transposon-mediated gene transfer, similar to our previous studies to derive iPSC-derived CAR-expressing NK cells that have now been translated into clinical trials. We used an anti-mesothelin (meso) scFv combined with 8 different CAR constructs with distinct intracellular signaling components. We found that the iPSC-Macs could express good levels of the CARs (iPSC-CarMacs). Function was again tested in vitro by phagocytosis of the Meso+ A1847 ovarian cancer cells. The iPSC-CarMacs with a Bai1 stimulatory domain consistently demonstrated the best activity in this assay system. We next tested the anti-meso-iPSC-CarMacs in vivo using the A1847 cells. Again, we demonstrate the iPSC-CarMacs combined with anti-CD47 mAb mediate significantly improved anti-tumor activity using this in vivo model compared to the non-CAR-iPSC-Macs + anti-CD47, p <0.005 (Figure). Survival studies are still ongoing.
Together, these studies demonstrate that iPSCs can be used to routinely and efficiently derive macrophages with potent anti-tumor activity. Additionally, CARs that are optimized for macrophage-mediated activity can be expressed to generate iPSC-CarMacs that effectively kill tumor cells in vitro and in vivo. These iPSC-CarMacs provide another approach to provide a standardized, targeted, off-the-shelf cell therapy product that can be used to treat both hematological malignancies as well as diverse solid tumors.
Kaufman: Shoreline Biosciences: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Research Funding; Qihan Biotech: Consultancy, Current holder of stock options in a privately-held company; VisiCELL Medical: Consultancy, Current holder of stock options in a privately-held company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal